May is Healthy Vision Month - click here for FREE healthy vision tips
Can-C Ingredients and Instructions

Can-C™ Eye Drops – Approved by Innovative Vision Products”
Two (5ml) vials per box
Can-C™ acts as both a stabilizer and carrier for safe delivery into the aqueous humor of the eye. Once delivered into the aqueous humor this bio-identical molecule becomes highly active.
Can-C™ Ingredients:
- Glycerin (lubricant) 1.0%
- Carboxymethylcellulose sodium (lubricant) 0.3%
Excipients:
- N-Acetyl-Carnosine (NAC) 1.0% (approved by IVP)
- Sterile Water (Ophthalmic Grade Isotonic Solution, pH 6.3 to 6.5)
- Buffers: Potassium Bicarbonate and Boric Acid
- Purified Benzyl Alcohol (Preservative) 0.3%
Dosage: Apply 4 drops daily to each affected eye.
Each carton of Can-C™ contains two (5ml vials) and each 5ml vial contains approximately 65-drops. Applying two drops to each eye, morning and evening, one box of Can-C™ will last approximately three weeks. (NOTE: Will last 6 weeks if treating one eye)
- (3 week supply) for both eyes = 1 box
- (2 month supply) for both eyes = 3 boxes
- (4 month supply) for both eyes = 6 boxes
- (6 month supply) for both eyes = 8 boxes
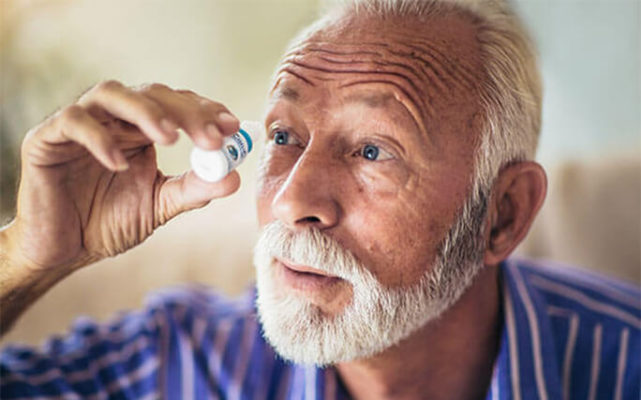
Eye Drop Application Instructions:
- Wash your hands with soap and water.
- Look toward the ceiling with both eyes open.
- Pull lower lid down with one hand to enlarge the opening.
- Hold bottle in other hand and turn directly upside down. Then carefully line up with the eye.
- Gently squeeze to instill “one drop” then gently close the eye for 60 seconds. The tip of the vial should never touch the eye.
- Wait 5 – 10 minutes or even an hour or more between applications to the same eye to prevent overflow of the product and to ensure maximum benefit.
- For cataract, apply a total of 4 drops daily to each affected eye. For general health improvement apply 2 drops daily to each eye.
Instructions on applying drops to dogs: click here for article
Storage: Keep in cool dry environment and do not expose to extreme temperatures. All open vials should be discarded after 30 days.
Can-C™is Manufactured in a GMP (Good Manufacturing Procedures) certified, pharmaceutical facility which meets ISO 9001:2000 and ISO 13485:2003 standards for the design/formulation and manufacture of sterile contact lens solution and pharmaceutical solutions per FDA guidelines. Only the lubricants (Carboxymethylcellulose sodium) and (glycerin) are approved by the FDA for ophthalmic use, and can be listed as active ingredients on our label. Can-C™ is not FDA approved for the treatment of cataracts therefore N-Acetylcarnosine (1%) is listed as an inactive ingredient on the label.
Disclaimer: The above information is presented for educational purposes only and does not replace the advice of your physician.

