April is Women's Eye Health and Safety Month - click here for FREE healthy vision tips
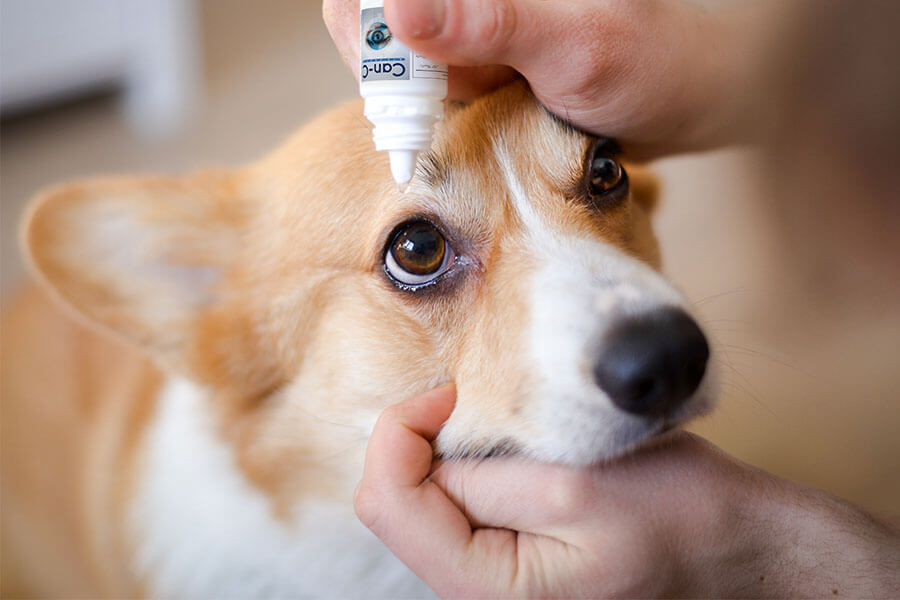
Can-C Eye Drops for Dogs
- 1 month supply
Can-C Batch Expiration Date:
October 31, 2027
$49.00 — or $49.00 Original price was: $49.00.$44.10Current price is: $44.10. / month
- Supports eyes’ natural healing process*
- Soothing Lubricating Eyedrops*
Initial results in only 3 months time!
Optimal Results within 6 – 12 months!
Two (5ml) vials per box.
Based on applying 3 drops daily to each eye, 1 box = 4 week supply.Click here to BUNDLE & SAVE
Based on applying 3 drops daily to each eye, 1 box = 4 week supply.Click here to BUNDLE & SAVE
- Worldwide Shipping
- Free shipping on all U.S. orders!
Can-C™ Ingredients:
- Glycerin (lubricant) 1.0%
Excipients:
- N-Acetyl-Carnosine (NAC) 1.0% (approved by IVP)
- Carboxymethylcellulose sodium (lubricant) 0.3%
- Sterile Water (Ophthalmic Grade Isotonic Solution, pH 6.3 to 6.5)
- Buffers: Potassium Bicarbonate and Boric Acid
- Purified Benzyl Alcohol (Preservative) 0.3%
Safe & Effective Ingredients
The healthy eye contains high levels of L-Carnosine. Interestingly, the eyes of those suffering from degenerative ocular diseases such as cataracts were found to be grossly deficient in this essential ocular antioxidant. Can-C™ replenishes the eye’s natural levels of this potent, natural antioxidant.
Enhancing the Eye's Restorative Properties
One of the most amazing features of the eye is its ability to repair itself and prevent infection after injury. Can-C™ drops support the healing process by keeping the eye well lubricated and delivering essential nutrients to support the eyes’ natural healing process.

Bundles containing Can-C Eye Drops for Dogs
Pets 90 Day Bundle
3
3 month supply
Take care of your best friend’s eyes
$150.00 one-time or
$142.50 every 3 months


